
If these values do indeed reflect the actual sizes of the atoms, then we should be able to predict the lengths of covalent bonds formed between different elements by adding them. In a similar approach, we can use the lengths of carbon–carbon single bonds in organic compounds, which are remarkably uniform at 154 pm, to assign a value of 77 pm as the covalent atomic radius for carbon. The van der Waals radius is half the distance between chlorine nuclei in two different but touching Cl 2 molecules. The covalent radius of Cl 2 is half the distance between the two chlorine atoms in a single molecule of Cl 2. (d) This is a depiction of covalent versus van der Waals radii of chlorine. (c) The van der Waals atomic radius, r vdW, is half the distance between the nuclei of two like atoms, such as argon, that are closely packed but not bonded. 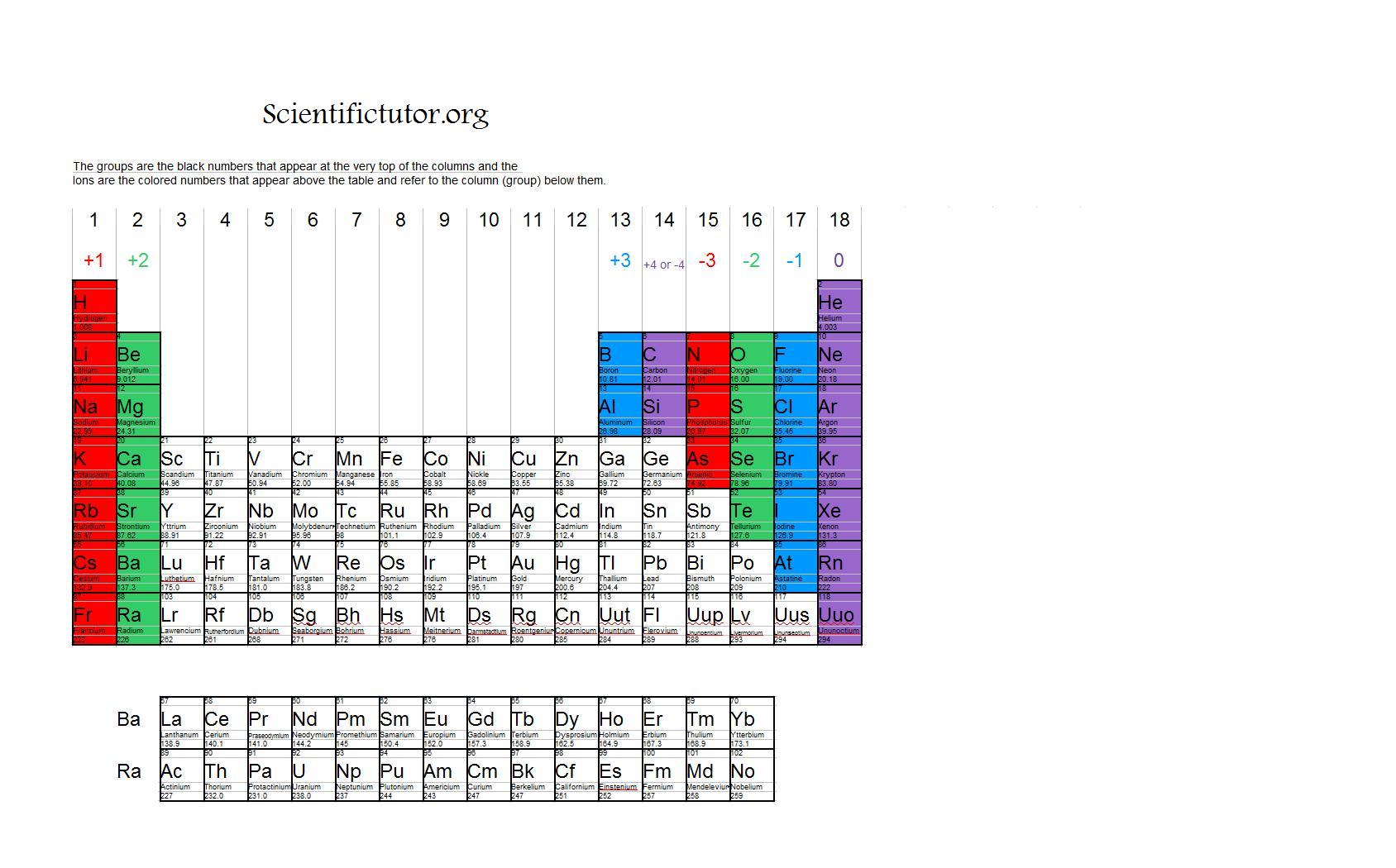
(b) The metallic atomic radius, r met, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as aluminum. (a) The covalent atomic radius, r cov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. You should remember that ionic bonds are normally strong, but they are very weak in water.\): Definitions of the Atomic Radius. Now you have sodium (Na +) and chlorine (Cl -) ions floating around the solution. It happens easily because of the electrical attraction of the water. However, if you put that salt into some water (H 2O), the bonds break very quickly. It would be nearly impossible to break those ionic/electrovalent bonds. Salt is a very strong bond when it is sitting on your table. Look at sodium chloride (NaCl) one more time. 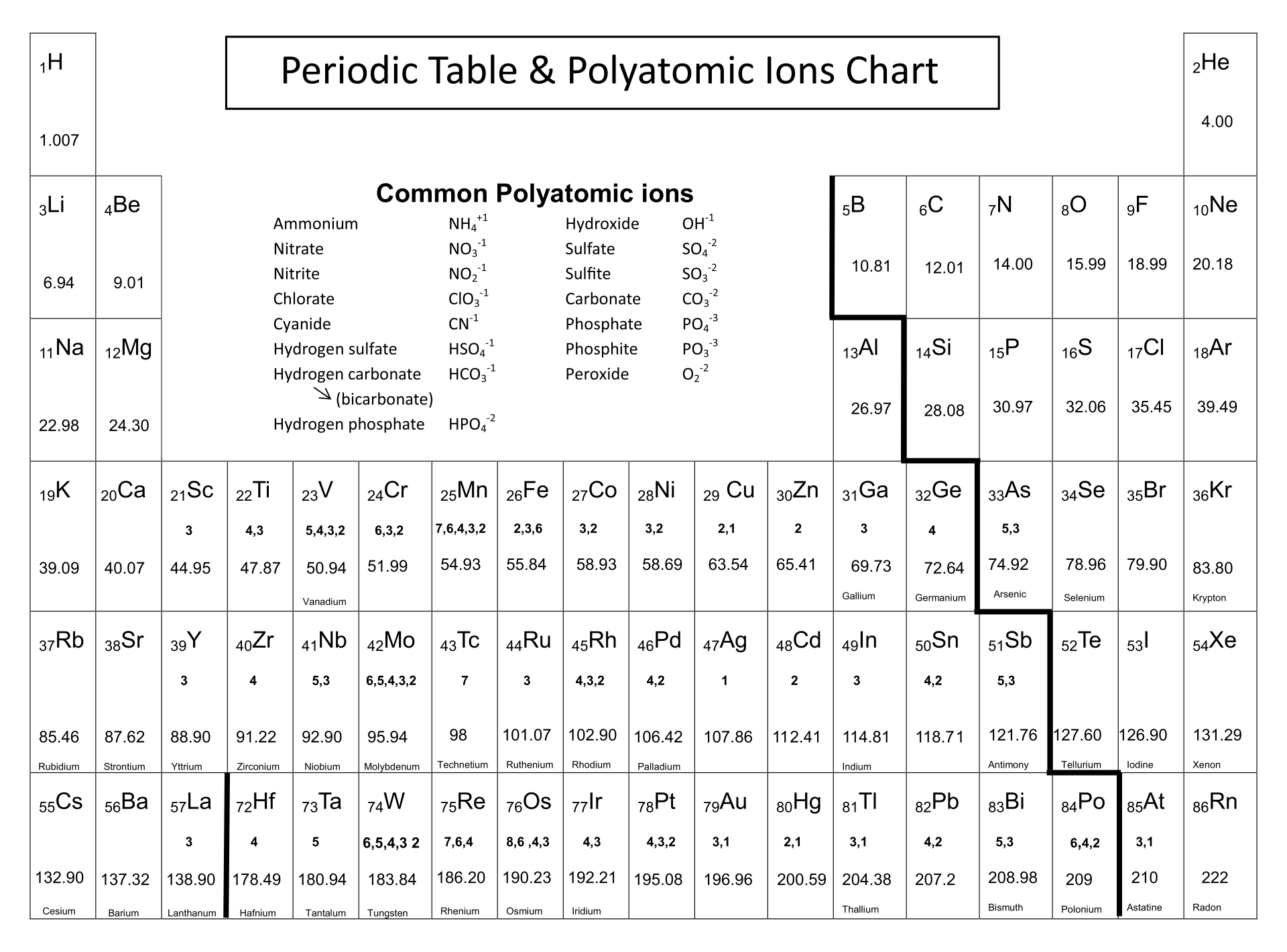
Sodium and chlorine ions alone have a very strong bond, but as soon as you put those ions in a solution with H +, OH -, F - or Mg ++ ions, there are charged distractions that break the Na-Cl bond. Scientists call these groups " ionic agglomerates." When in the presence of other ions, the electrovalent bonds are weaker because of outside electrical forces and attractions. They are just groups of charged ions held together by electric forces. You may have heard of the term "ionic bonds." Ionic bonds are electrovalent bonds. There are two main types of bonding, covalent and electrovalent. It is also a measure of how many electrons are excited about bonding with other atoms. Valence is a measure of how much an atom wants to bond with other atoms. That trend means that the left side has a positive valence and the right side has a negative valence. If you look at the periodic table, you might notice that elements on the left side usually become positively charged ions (cations) and elements on the right side get a negative charge (anions). Electrovalence is just another word for something that has given up or taken electrons and become an ion. Almost any ion with a negative charge will be interested in bonding with you.ĭon't get worried about the big word. Other electrically charged atoms (ions) of the opposite charge (negative) are now looking at you and seeing a good partner to bond with. You lost an electron and you are "happy." So what makes you interesting to other atoms? Now that you have given up the electron, you are quite electrically attractive. Your whole goal as an atom was to become a "happy atom" with completely filled electron shells. As an ion you are now something completely new. Since you're missing an electron, you aren't really a complete sodium atom either. That's the same number of electrons as neon (Ne). You have one less electron than your atomic number. You are still the element sodium, but you are now a sodium ion (Na +). That missing electron gives you a positive charge. You are also an ion and missing one electron. Chlorine has seventeen electrons and only needs one more to fill its third shell and be "happy." Chlorine will take your extra sodium electron and leave you with 10 electrons inside of two filled shells. Whenever an atom has full shells, we say it is "happy." Let's look at chlorine (Cl). When you lose that electron, you will you’ll have full shells. You need to find another element that will take that electron away from you. What do you do if you are a sodium (Na) atom? You have eleven electrons - one too many to have an entire shell filled. When you have an extra electron or two, you have a negative charge. When you are missing an electron or two, you have a positive charge. Ions are atoms with extra electrons or missing electrons. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. A normal atom has a neutral charge with equal numbers of positive and negative particles. The atomic number of an element, also called a proton number, tells you the number of protons or positive particles in an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
